Health
FDA Official’s Allegation of Vaccine-Linked Child Deaths Sparks Controversy

The recent assertion by the Food and Drug Administration’s (FDA) top vaccine regulator, Dr. Vinay Prasad, claiming that the COVID-19 vaccine has caused at least 10 deaths in children, has raised significant concerns among experts in the medical community. In an email sent to FDA staff on October 27, 2023, Prasad called for a reevaluation of how vaccines are regulated, positioning this assertion as a pivotal moment for the agency.
Prasad’s memo declared that regulatory standards for new vaccines would be intensified in light of these alleged deaths, stating, “This is a profound revelation.” He emphasized that the FDA would now officially recognize the potential lethal impact of COVID-19 vaccines on American children, a statement that has drawn scrutiny due to a lack of detailed supporting data. Experts, including public health officials and vaccine researchers, expressed skepticism regarding the memo’s claims, describing them as “extraordinary” and demanding further evidence.
The FDA has not clarified its next steps following Prasad’s announcement. The memo lacks detailed information on the alleged deaths or plans for further investigation, leading to uncertainty about how the agency intends to address the issue. For instance, while Prasad indicated a need for more conclusive data regarding vaccines administered during pregnancy, he did not specify which vaccines were involved in his claims.
Some critics have pointed out that the memo includes inaccuracies, such as the implication that the federal government dictates school vaccine mandates. Dr. Michael Osterholm, director of the University of Minnesota’s Center for Infectious Disease Research and Policy, described the claims as “irresponsible science” that could mislead the public. He suggested that the FDA refer the matter to the National Academies of Sciences, Engineering, and Medicine for an independent review.
Several anonymous FDA staff members have voiced their concerns about the memo, labeling it misleading and politically charged. The internal dissent reflects a broader apprehension regarding the implications of linking vaccine policies to potentially unverified claims. Paul Offit, a professor at Children’s Hospital of Philadelphia and co-developer of a vaccine for rotavirus, criticized the vagueness of the proposed policy changes, stating, “It’s an extraordinary claim and should be backed by extraordinary evidence.”
In his email, Prasad referenced ongoing investigations into reports from the Vaccine Adverse Event Reporting System (VAERS), focusing on 96 reported deaths from 2021 to 2024. He asserted that “no fewer than 10 are related” to the COVID-19 vaccine, although this conclusion remains contested among experts who question the attribution methodology.
Experts like Daniel Jernigan, former director of the CDC’s National Center for Emerging and Zoonotic Infectious Diseases, have called for greater transparency regarding the data used to support such claims. They argue that a detailed analysis is essential to confirm any causal relationships between vaccination and reported deaths.
Prasad’s memo also suggested changes to the regulatory framework surrounding vaccines, indicating that the FDA may reassess how multiple vaccines are administered concurrently. Concerns have been raised that such changes could disrupt existing childhood vaccination schedules, potentially leading to lower vaccination rates.
The memo’s implications extend beyond the immediate controversy, as it touches on broader discussions about the risks of COVID-19 to children. Although Prasad characterized the risk of death from COVID-19 as “tremendously low,” data from a 2023 JAMA paper indicated that 821 individuals aged 19 and under died of COVID-19 in the year ending July 2022, making it one of the leading causes of death in that age group.
Amid ongoing discussions about vaccine safety and efficacy, experts continue to emphasize the necessity of rigorous scientific evaluation. As the FDA prepares for its upcoming meetings, including a key advisory committee session, the stakes remain high for public health policy and the future of vaccination programs. The call for a more robust evidentiary basis for vaccine regulation reflects a growing demand for accountability and transparency in health policy decisions.
As the situation evolves, many in the medical field will be watching closely to see how the FDA addresses these claims and whether further evidence will be presented to substantiate Prasad’s assertions.
-

 Top Stories2 months ago
Top Stories2 months agoNew ‘Star Trek: Voyager’ Game Demo Released, Players Test Limits
-

 World2 months ago
World2 months agoGlobal Air Forces Ranked by Annual Defense Budgets in 2025
-

 World2 months ago
World2 months agoMass Production of F-35 Fighter Jet Drives Down Costs
-
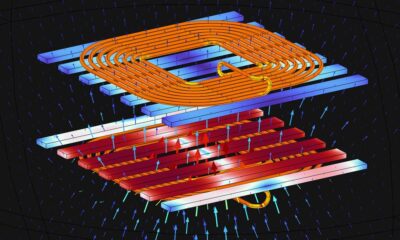
 World2 months ago
World2 months agoElectrification Challenges Demand Advanced Multiphysics Modeling
-
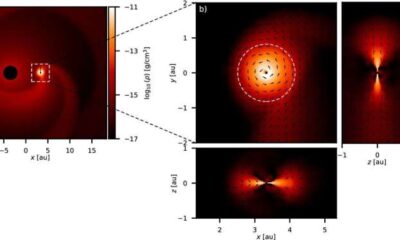
 Science2 weeks ago
Science2 weeks agoALMA Discovers Companion Orbiting Giant Red Star π 1 Gruis
-

 Business2 months ago
Business2 months agoGold Investment Surge: Top Mutual Funds and ETF Alternatives
-

 Science2 months ago
Science2 months agoTime Crystals Revolutionize Quantum Computing Potential
-

 Top Stories2 months ago
Top Stories2 months agoDirecTV to Launch AI-Driven Ads with User Likenesses in 2026
-

 Entertainment2 months ago
Entertainment2 months agoFreeport Art Gallery Transforms Waste into Creative Masterpieces
-

 Business2 months ago
Business2 months agoUS Government Denies Coal Lease Bid, Impacting Industry Revival Efforts
-

 Health1 month ago
Health1 month agoGavin Newsom Critiques Trump’s Health and National Guard Plans
-

 Lifestyle1 month ago
Lifestyle1 month agoDiscover Reese Witherspoon’s Chic Dining Room Style for Under $25